At Reed Tech Life Sciences, we know the challenges of locating and securing important documentation for both internal and external downstream users. In a recent webinar presentation, Reed Tech Alliance member, Greenlight Guru, showcased how their solution empowers customers, specifically the medical device market, to structure documentation with flexible visibility and security. We walked away with a few key takeaways that may be beneficial to your company and its documentation users.
Scale Documentation within an Organization
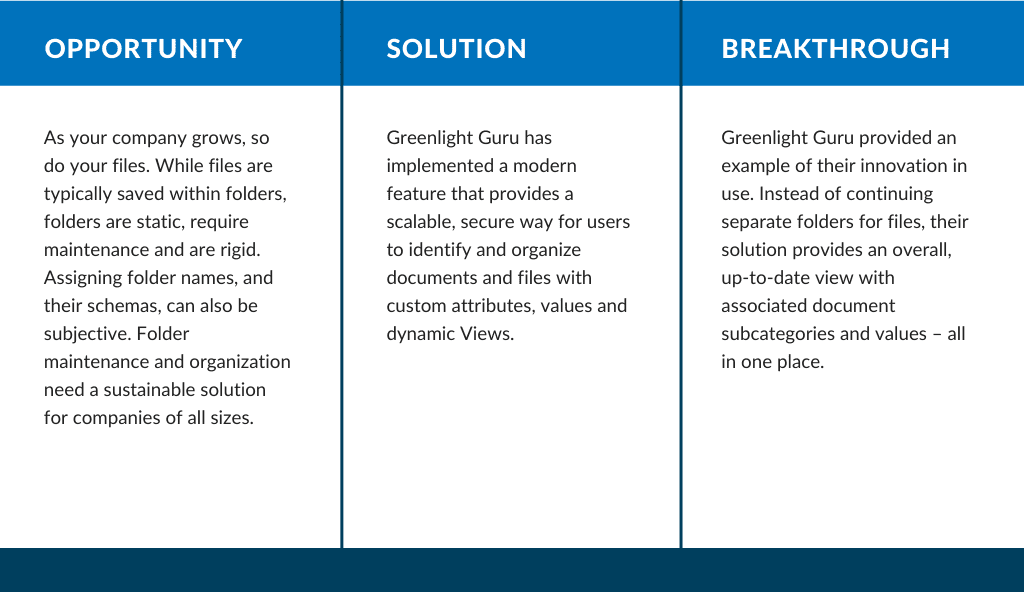
The challenges in document management visibility are complex. For instance, companies can become quickly buried under several folders, containing multiple versions of various files. Greenlight Guru listened to this market feedback and created a solution for a flexible document organization.
Improve Structure for Document Organization
Medical device companies need to be able to find documents easily (for impending regulation compliance, audits, etc.) yet structure is limited and searchability can be narrow. One of the most important key takeaways from Greenlight Guru’s new feature, Flexible Document Organization, is the ability to customize within the Document Workspace. This will allow users to easily store, search and locate documents.
Through Flexible Document Organization, users can:
-
-
- Customize the Document Workspace to meet changing needs within your organization
- Create custom terms (Attributes and Values) to identify documents
- Save and share Views that automatically update as soon as documents are uploaded and updated
-
Apply the Flexible Document Organization Feature
A use case noted by a Greenlight Guru customer explained, “instead of creating separate views for ‘Meeting Minutes – Internal’ and ‘Meeting Minutes – External’ can we have an overall view for Meeting Minutes and subcategories like Internal, External?” Greenlight Guru listened to this feedback and established the Flexible Document Organization feature to provide an inclusive Document Workspace, supporting dynamic Views, and custom Attributes and Values.
To best understand and visualize how Flexible Document Organization can be of service to your organization, Greenlight Guru offers a Live demo. Connect with Greenlight Guru and see how your current document management system can improve and scale within your company.
About Greenlight Guru
Greenlight Guru is the only dedicated Medical Device Success Platform (MDSP) designed specifically for medical device companies. The platform helps companies bring safer products to market faster, simplifies FDA and ISO regulatory compliance and provides a single source of truth by connecting the management of all quality processes like CAPAs, risk, audits and more. Greenlight Guru’s platform is used by thousands of organizations across the globe to push beyond baseline compliance and achieve True Quality for their medical devices. For more information, visit https://www.reedtech.com/referrals-to-reed-tech-gg/.