Are makers of eyewear products subject to Unique Device Identification (UDI) requirements for compliance with EUDAMED? Recent news would indicate the answer is ‘yes’. For those manufacturers of eyewear lenses and frames, a recently posted guidance document from the EU Medical Device Coordination Group (MDCG) defines the application of UDI to Spectacle Lens and Ready Readers.
Summary
- Spectacle Lens – glass or plastic articles that apply optical correction to vision
- Spectacle Frame – supporting “arms” holding the lens in position on the head
- Spectacles – assembly containing the spectacle lens and spectacle frame, aka, glasses
- Ready Readers – OTC glasses correcting near-sighted conditions, aka, ‘readers’ (in US)
Ready readers or ready-made reading spectacles’ are spectacles that have 2 single vision lenses each of which have the same positive spherical power not exceeding 4 dioptres and the purpose of which is to help with near vision tasks. These “ready-readers” are widely available across Europe and can be bought without prescription from a variety of outlets, without professional advice or supervision.
This short four-page MDCG document provides grouping criteria and attributes to assign Basic UDI-DI and UDI-DI identifiers to both Spectacle Lens and Ready Readers. By implication, these two medical device categories must comply with MDR UDI regulations, e.g., UDI & device registration to EUDAMED and UDI labeling.
The guidance document does not mention any exception to the MDR regulations or extended compliance timelines. It also does not indicate the use of the listed attributes – are the 3 to 6 defined attributes used only for logically grouping products or to be included in the UDI data record submitted to the EUDAMED UDI module or perhaps some other use? Though not comprehensive, this MDCG document begins to address EU UDI compliance to eyewear products, see table below.
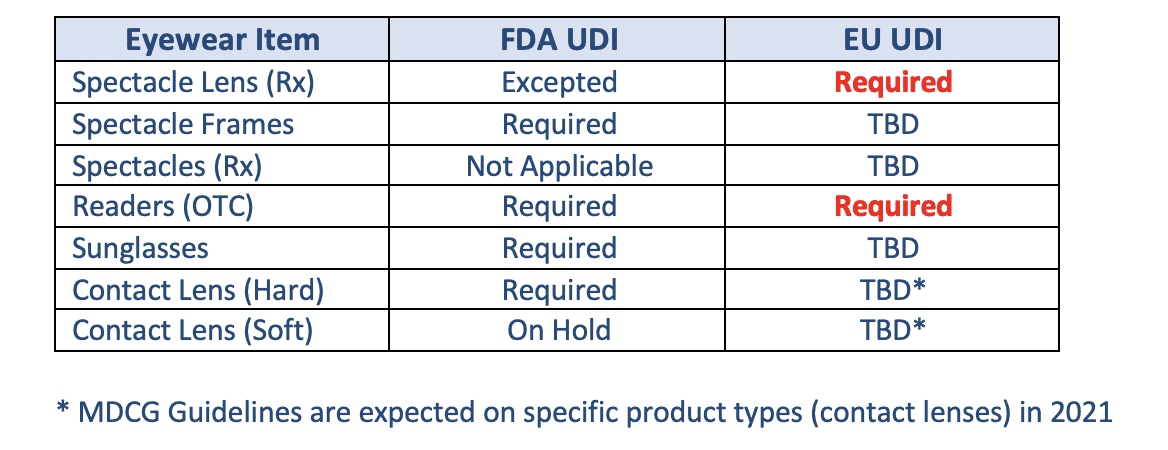
Potential Impacts
If the above interpretation is confirmed, the EU requirement for Spectacle Lens to comply with UDI is a direct opposite position from the US FDA decision to except Spectacle Lens from UDI. It appears the EU decision would impact makers of Spectacle Lenses, requiring UDI records to be reported to EUDAMED.
Navigating the complexities of regulatory requirements for UDI for the US FDA or other global health authorities can quickly become a burden.
For more information, contact us at [email protected] or call +1-215-557-3010
Source: MDCG 2020-18, MDCG Position Paper on UDI assignment for Spectacle lenses & Ready readers
