Medical Device Product Data Management
Reed Tech SingleSource™ for Medical Devices
How can Reed Tech SingleSource™ for Medical Devices help manage the complexities of UDI for multiple global health authorities?
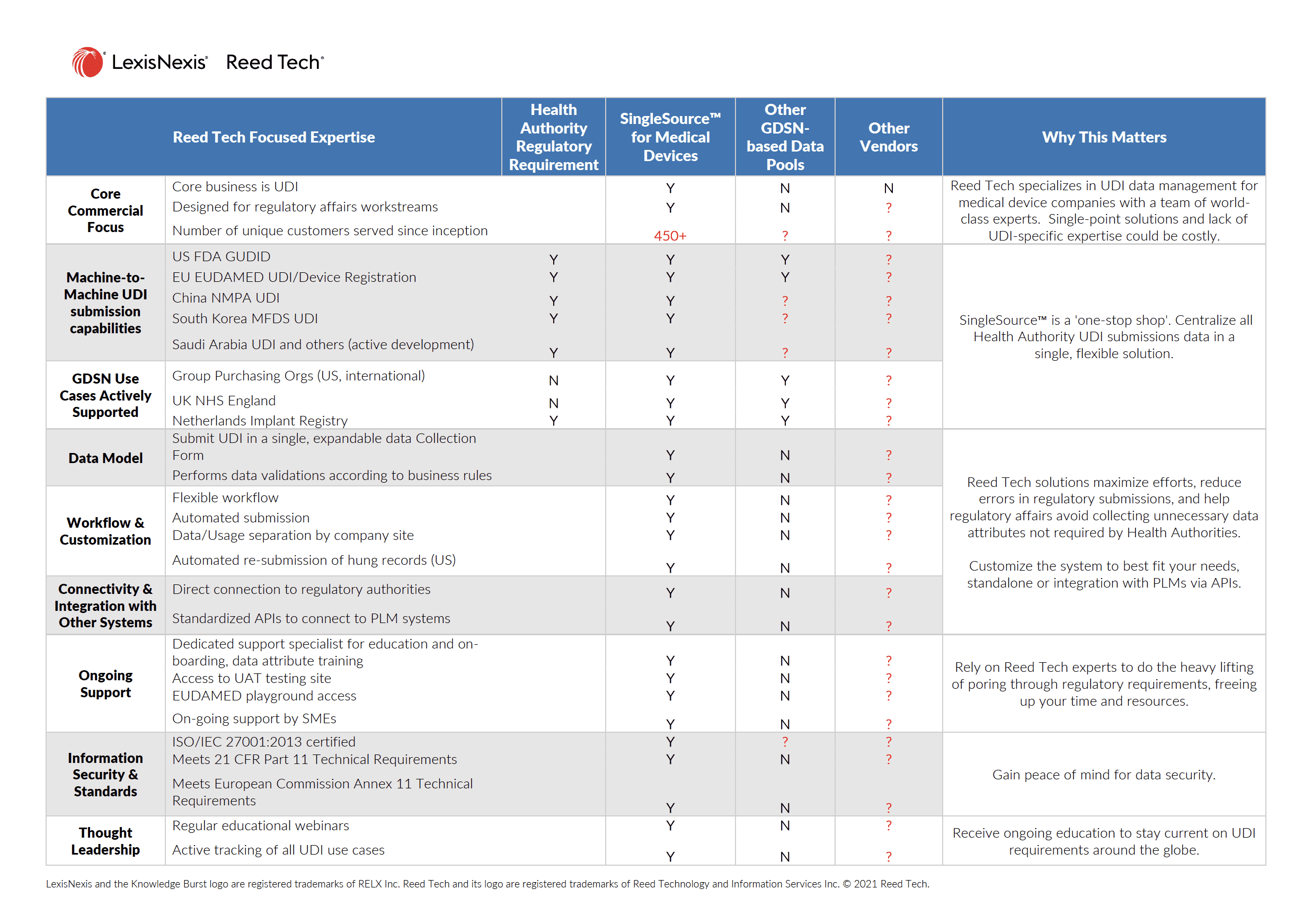
Our core business is managing and submitting Unique Device Identification (UDI) data. Starting with the US FDA, medical device regulators are instituting UDI compliance requirements in regional markets around the world. Numerous Health Authorities, including the European Commission (EUDAMED), China NMPA, South Korea MFDS and Australia TGA/AusUDID have issued regulations with more to follow.
Medical device manufacturers are finding that UDI implementation can be difficult. The regulations are new, with varying levels of clarity and they are not harmonized. They have differing legislation, policy, timing, data and submission approaches. For teams managing UDI regulatory data, the preparation, collection, submission and maintenance details can quickly become a complex and overwhelming task.
SingleSource™ centralizes UDI submissions data in a single, flexible solution. Start with the regions your devices are marketed in and that accommodate machine-to-machine submissions. Activate and pay for channels when you enter a market, not assumed within a bundled rate. Plus, receive training and continual UDI educational opportunities as new information is published by Health Authorities.
What can Reed Tech SingleSource™ for Medical Devices do for your team?
The Challenge
Achieving Compliance—For all the health authority channels with regulatory requirements, engage with UDI expertise with a single, scalable, and flexible solution to collect, maintain, validate, and submit product data.
Utilizing an In-Country Legal Representative—Assign roles/responsibilities for corporate users and local affiliates for a shared team view into UDI data. Avoid multiple costs and provide transparency.
Maximizing Efforts—One ‘source of truth’ is needed to comply with health authority channels, anticipating differing product attributes, data validation rules and submission standards by country.
Subject-Matter Expertise—to navigate, understand, and react is a necessity.
Sharing Consistent Data—Commercial Trading Partners within the supply chain will require the same product data set used for regulatory submission either via the global data pool or other means.
Trusted Expertise—The electronic submission process and business rules carry nuances from region to region.
Cost Predictability—Protect your budget with a single solution when new regulatory authorities come into the picture. Ensure business continuity is achieved with minimal disruption.
Learn more about approaches to managing UDI product data
SingleSource™ for Medical Devices Features
Speak to a Subject-Matter Expert
SingleSource™ for Medical Devices was built to meet the product data management needs of Medical Device manufacturers. See what it can do for you.
Sign up for a demo today.

“We were guided through the process step by step, and when we encountered an issue it was resolved promptly.”