LexisNexis Reed Tech
Knowledge Center
Explore our library of blogs, short videos, virtual event recordings and training topics
Recent Blogs
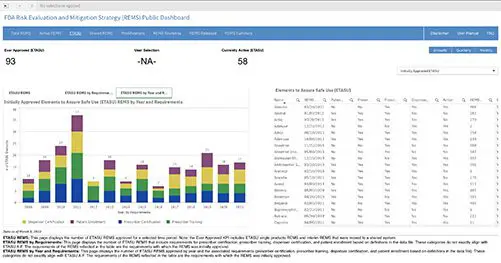
Public Dashboard Now Available for REMS Data
A new public dashboard has been launched by the Food and Drug Administration (FDA) for public access to data for drugs...
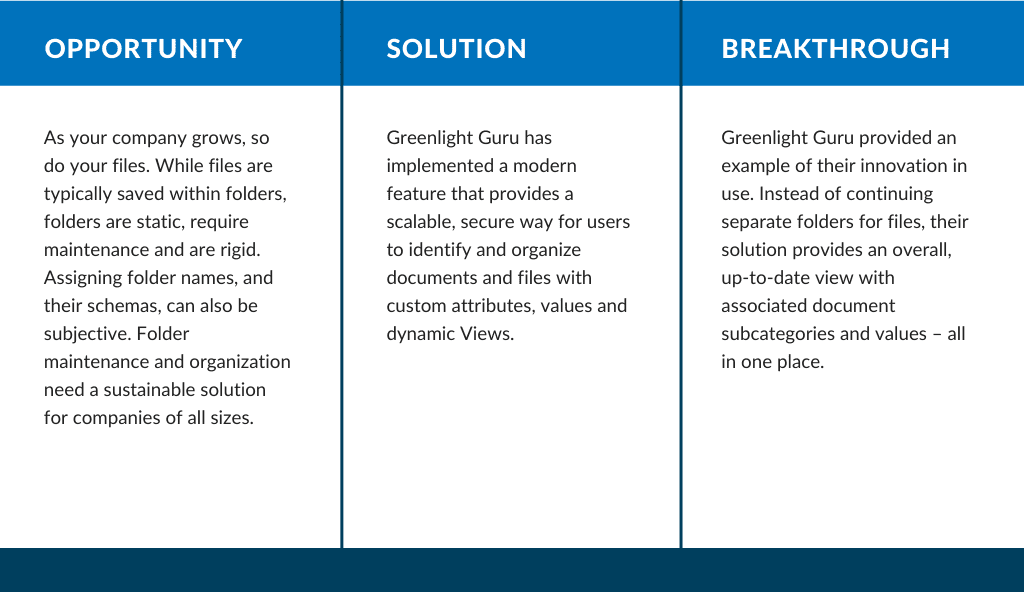
Organizing and Finding Documents Easily for Medical Device Companies, Featuring Greenlight Guru
At Reed Tech Life Sciences, we know the challenges of locating and securing important documentation for both internal...
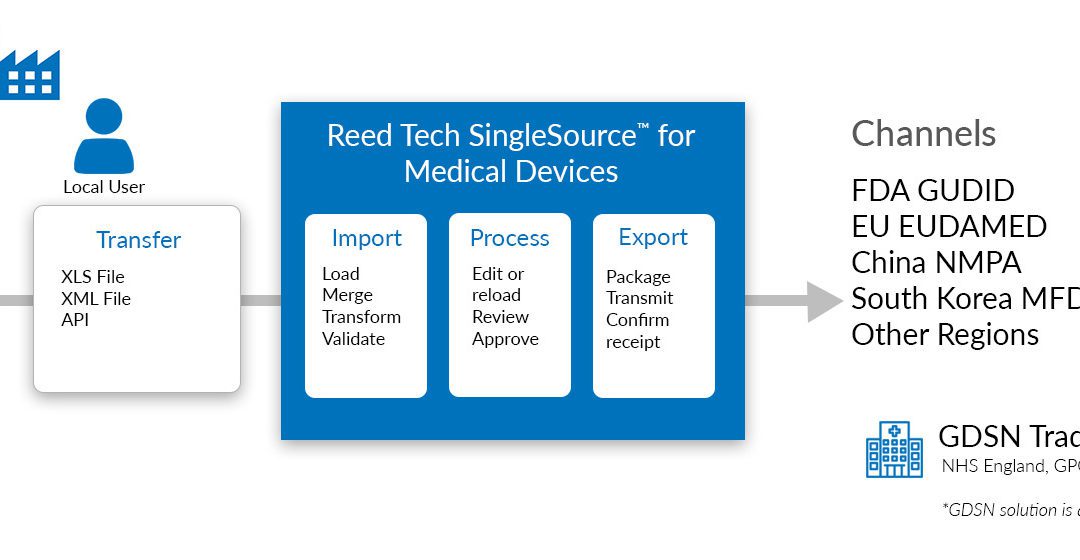
How Can You Prepare for Unique Device Identification Product Data Submission in Asia?
Reed Tech subject matter experts, Gary Saner, Information Solutions Senior Manager and Patti Shragher, Medical Device...

LexisNexis® Reed Tech and Greenlight Guru Announce Strategic Alliance to Guide Customers to Market Faster
LexisNexis® Reed Tech has teamed up with Greenlight Guru, the leading Medical Device Success Platform for medical...
How Does Software as a Medical Device Relate to Unique Device Identification?
Determining Software as a Medical Device is essential for regulatory compliance. This involves classifying software as a Device Component, Accessory, or Standalone Device, and understanding FDA’s UDI requirements and the implications of software changes on device identification.

Is My Product a Medical Device?
A frequent question on medical devices concerns determining ‘if’ a product is defined by US FDA as a ‘medical device’....
