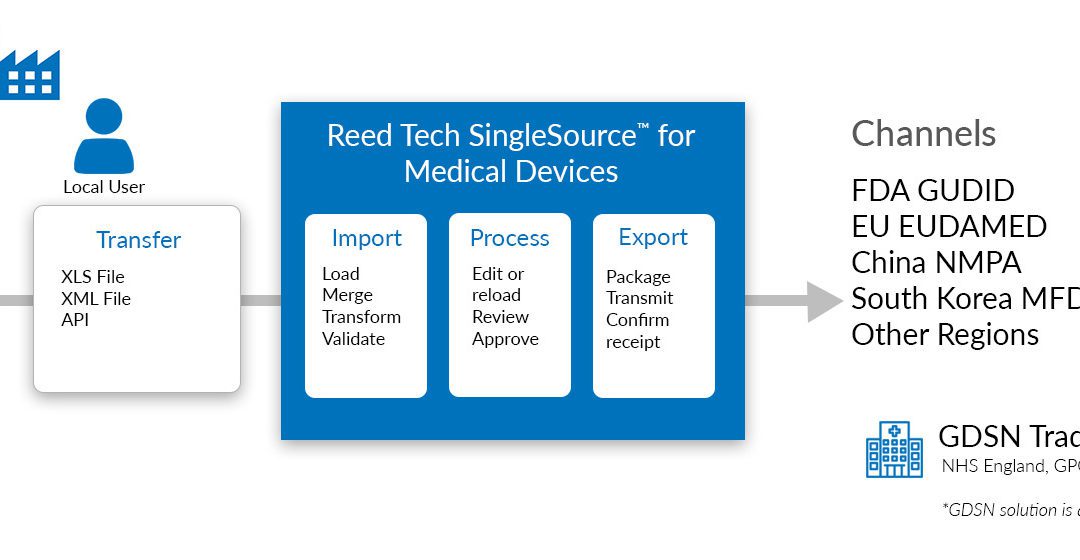
Reed Tech subject matter experts, Gary Saner, Information Solutions Senior Manager and Patti Shragher, Medical Device Senior Account Executive and Team Lead, host a webinar discussion dedicated to Unique Device Identification (UDI) requirements for health authorities...