The Modernization of Cosmetics Regulation Act of 2022 overhauls existing regulations, mandating FDA-established Good Manufacturing Practices for cosmetics manufacturers.

LexisNexis Reed Tech
Explore our library of blogs, short videos, virtual event recordings and training topics

The Modernization of Cosmetics Regulation Act of 2022 overhauls existing regulations, mandating FDA-established Good Manufacturing Practices for cosmetics manufacturers.

As of December 28, 2022, all new REMS (Risk Evaluation and Mitigation Strategy) submissions and all REMS updates will be required to be submitted in SPL (Structured Product Labeling) format per the FDA. This is in compliance with the Final Guidance on REMS issued by FDA in late December 2020. This means that any current REMS registrations will not need to be submitted in SPL until they require updates. There are currently 62 active REMS—58 ETASU (Elements to Assure Safe Use) and 4 basic (non-ETASU.)
Continue to be at the forefront of regulatory chatter. Our customers, both primarily medical device and primarily pharmaceutical, are regularly coming to us with questions surrounding this topic. Reed Tech answers these questions here in this blog post

Reed Tech recently presented a webinar to update customers with the most recent information about XML product...

Reed Tech habitually attends industry conferences and forums to share our expertise and gain knowledge from our...

Discover LexisNexis Reed Tech and Schlafender Hase’s alliance for streamlined life sciences data management and regulatory solutions.
Recently, Reed Tech subject-matter experts, Gary Saner and David Wilson, hosted a webinar to provided a recap of what this new deadline means for pharma companies and how to ensure compliance, as well as explained the implications of recent updates and the progress of the draft guidance.
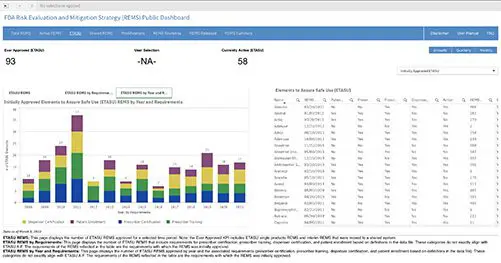
A new public dashboard has been launched by the Food and Drug Administration (FDA) for public access to data for drugs...

The Food and Drug Administration (FDA) has informed industry that the February 15, 2022 date for submitting 2020 data per Reporting Amount of Listed Drugs and Biological Products Under Section 510(j)(3) of the Federal Food, Drug, and Cosmetic Act, originally referred to as a deadline, will now be the recommended date.